Research
Alternating Weekly Treatment Schedule for Superficial Radiotherapy…
ALTERNATING WEEKLY TREATMENT SCHEDULE FOR SUPERFICIAL RADIOTHERAPY FOR NON-MELANOMA SKIN CANCERS
Tom J. Kim, William Ting, MD
California Dermatology Care, San Ramon, CA.
Introduction
Superficial radiotherapy, or superficial radiation therapy (SRT), is a painless, noninvasive option for treating nonmelanoma skin cancers (NMSCs) such as basal cell carcinoma (BCC), squamous cell carcinoma (SCC), or basosquamous cell carcinoma (BSC). These skin cancers typically manifest in patients of Caucasian descent, Fitzpatrick Type I – III, with increased risk above the age of 601 , 2. In order to treat an NMSC with SRT, radiation is administered to the surface layer of the skin, typically between 4500 to 5500 cGy divided into low dose fraction schemes. However, factors may alter the total radiation administered, such as size of the cancer, skin thickness, vascularity or underlying bone and cartilage. The typical schedule of treatment is SRT administered one to three times weekly3.
Background
Mohs micrographic surgery and surgical excision remain the primary treatment of NMSCs as procedural therapy, and are regarded as the first-line forms of treatment per clinical guidelines4. However, SRT offers an alternative treatment method for patients that are elderly, unable or unwilling to undergo surgery, have skin cancers located in regions difficult to surgically excise, are taking anticoagulant or other blood-thinning medications, or have had recurrence of skin cancer after attempted surgical removal5.
SRT is a safe and effective treatment for NMSC, and can offer superior cosmetic outcomes compared to surgical intervention due to limiting damage to the affected area. Therefore, in certain clinical contexts, it should be considered as a first-line therapeutic modality of NMSC6.
However, it is not without its drawbacks. Previous studies have determined that erythema and radiation dermatitis are common side effects, though these resolve over time7 . Additional studies have warned about the inflammation SRT causes which potentially impairs functionality of the site or limb administered8. Furthermore, rates of recurrence typically average around 1.9% at 2 years and 5% at 5 years, cumulatively9.
Objective
The purpose of this retrospective study was to determine the impact of radiation dermatitis, clinical cure rate, and cosmetic outcomes of patients on alternating weekly SRT treatment schedule vs. traditional weekly SRT treatment protocol which typically does not allow for more than 5 days of treatment gap between SRT fractions. Specifically, we want to examine whether the modified alternating SRT schedule of patients to be treated twice weekly every other week,
for a total of 17 fractionated sessions offer any treatment advantage over traditional/manufacturer recommended SRT treatment protocol of 2 to 3 times a week for 4-5 weeks straight.
Methods
Chart review of SRT patients treated for NMSC from ‘which date’ to ‘what date’ noted out of
690 NMSCs were treated from July 7, 2015 to October 31, 2020. Of these patients, 311 had prior history of BCC, 299 had prior history of SCC, 220 had no prior history of NMSC, 39 had prior history of malignant melanoma and 31 had prior history of an unspecified NMSC.
28 patients had undergone a schedule break or modification for reasons including but not limited to vacation, personal illness, quarantine due to COVID-19 outbreak or other emergency. In these cases, 2-3 fractions were added per case depending on the number of doses already received and the size and depth of the NMSC.
The instrument used was a superficial radiation therapy SRT-100+ [Sensus Healthcare]; the same instrument was used during the duration of this retrospective study and was calibrated (insert here). The average energy administered was 5029.62 cGy ± 218.52 with an average dose of 293.19 cGy ± 6.84. All patients in this study had a 2 week follow-up after their final SRT dosage. 493 Caucasian patients (52.3% male, 47.7% female; average age 70.1 years ± 13.1) underwent SRT for 690 cases of NMSCs, from 2015 to 2020, using this alternating weekly treatment schedule with an average of 17.1 fractions.
Results
Table 1: Frequency of appearance of various NMSCs on the human body (n = 690).
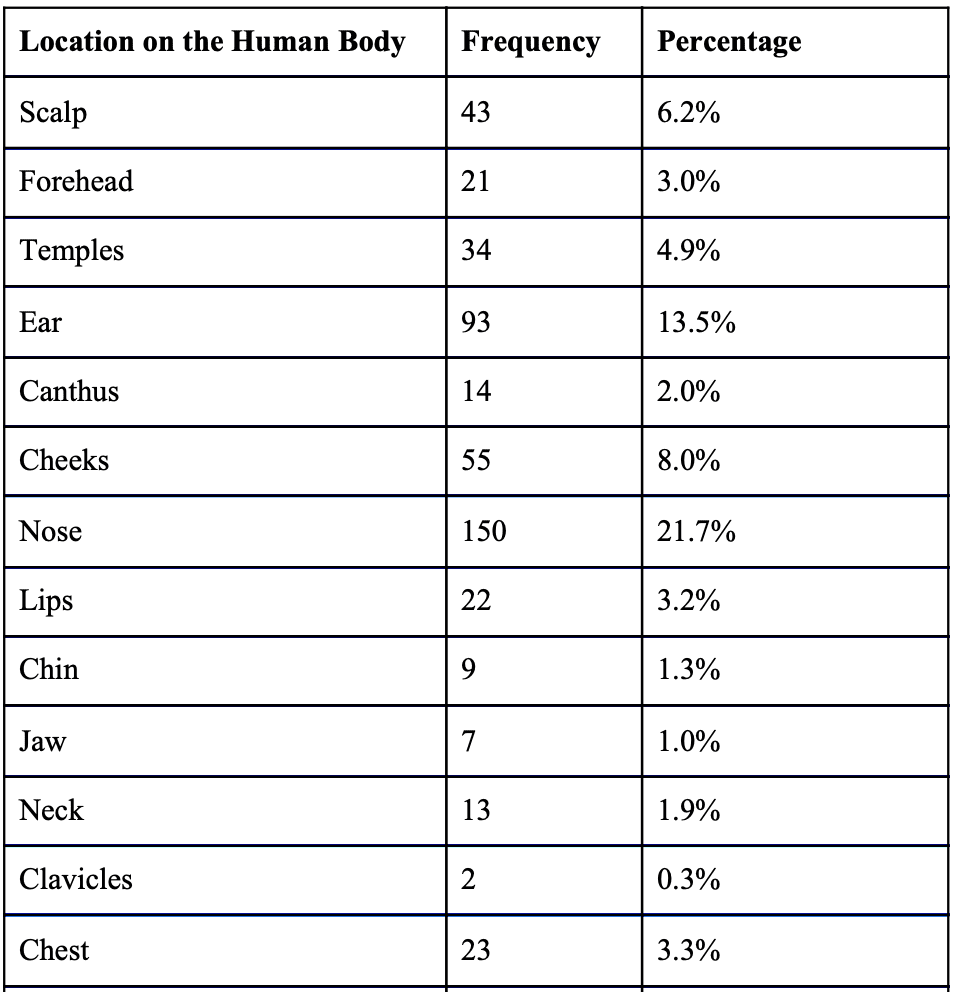
Of the 690 cases treated: 336 (48.7%) had histologically proven BCC (98 nodular, 27 invasive, 6 infiltrating, 5 superficial, 9 recurrent from MOHS or excision); 314 (45.5%) had SCC (128 in-situ, 41 invasive, 2 recurrent from MOHS, 1 ulcerating); 36 (5.2%) had BSC; 2 (0.3%) had microcystic adnexal carcinoma; 1 (0.01%) had atypical fibroxanthoma, and 1 (0.01%) had syringocystadenocarcinoma papilliferum.
Recurrence occurred in at least 7 (1.0%) patients, 5 male and 2 female; 3 BCC (2.13 years ± 0.89, 0.9cm ± 0.3), 1 BCC invasive (2.48 years, 0.6cm), 1 BCC nodular (4.2 years, 1.5cm), 1 BCC infiltrating (1.62 years, 0.5cm), and 1 SCC invasive (0.66 years, 1.8cm).
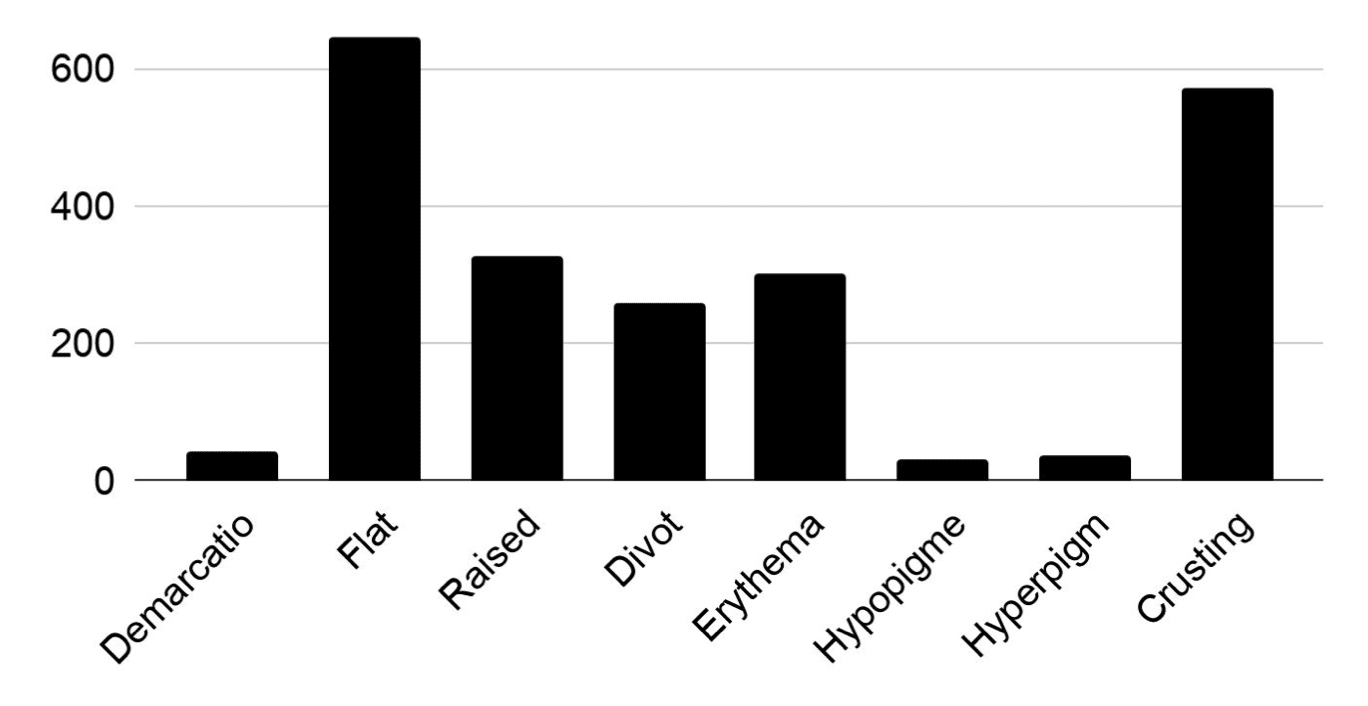
Figure 1: Common appearing side effects of SRT treatment
General Discussion
The presence of crusting is indicative of radiation dermatitis. Inflammation was typically presented as either being raised or erythematous if not both. High rates of radiation dermatitis (83.0%), divot (37.5%) and erythema (30.2%) were appreciated during treatment, but with lower rates of hyperpigmentation (5.4%), hypopigmentation (4.3%), or demarcation (6.2%) [Figure 1]. The presence of divoting in the skin, which starts to evolve over time in a 4-5 year period, has been a point of concern in SRT treatment; typically these are cosmetically rated poorly and are undesirable10. However the most common side effect of the treated NMSC was flattening of the lesion (93.6%) which persisted through subsequent follow-up visits.
Recurrence occurred in at least 7 (1.0%) patients, 5 male and 2 female; 3 BCC (2.13 years ± 0.89, 0.9cm ± 0.3), 1 BCC invasive (2.48 years, 0.6cm), 1 BCC nodular (4.2 years, 1.5cm), 1 BCC infiltrating (1.62 years, 0.5cm), and 1 SCC invasive (0.66 years, 1.8cm).
64.9% of patients had skin cancer on the face and scalp region, with the highest proportion of patients having a NMSC on the nose at 21.7% [Table 1] This presence of NMSC is typically indicative of overexposure to sunlight accumulated throughout the years11. While this data can be interpreted as that most NMSCs will appear on the, another valid interpretation is that patients are more likely to undergo SRT if the NMSC is on the face. It can be thus reasonably assumed that these patients would worry about the cosmesis regarding its treatment.
Future Direction
It is important to consider the patient’s clinical and social context in treatment of NMSC, particularly when it comes to use of SRT. Consideration must be made for optimal cure rate, impact on functional deficit, pain, and cosmetic outcome. It is unclear whether or not the modified alternating weekly SRT treatment schedule has any influence on the rate of recurrence. Additional confirmatory studies would be needed from other centers in the U.S. to ascertain our finding. In the near future, we would like to better understand how differing treatment schedule protocols affect clinical outcomes of SRT in the treatment of NMSC such as rate of recurrence of NMSC.
Limitations
This study was a retrospective study limited to one office in San Francisco Bay Area, and may not fully representative of other regions of the U.S. Furthermore we did not start photo documentation for each fractionated therapeutic from the initial 75 cases of the study cohort, from July 2015 to December 2016 (10.9% of cases), which limits documentation of side effects of SRT. Finally, although each patient had a 2-week follow-up after their SRT treatment, about 25% of patients did not return for follow-up; therefore we could not identify wound healing or recurrence in those patients.
Conclusion
SRT is a nonsurgical option for the treatment of NMSC which is traditionally administered 2 to 3 times weekly without planned treatment gaps. The modified alternating weekly treatment schedule which allows for planned treatment gaps of 10-12 days after 2 consecutive SRT fractionated sessions was designed to minimize radiation dermatitis and to provide better scheduling flexibility for patients undergoing SRT for NMSC. Compared to traditional SRT treatment schedule, the modified alternating weekly SRT treatment protocol resulted in less radiation dermatitis as well as improved cosmetic outcomes with lower rates of hyperpigmentation, hypopigmentation, and skin textural demarcation.
Acknowledgements
This project was supported by California Dermatology Care. We wish to recognize Iris Albert, R.T., the head SRT specialist who had administered these treatments and continued to collect the data that is present in this poster.
Citations
1. McGregor, S., Minni, J., & Herold, D. (2015). Superficial Radiation Therapy for the Treatment of Nonmelanoma Skin Cancers. The Journal of clinical and aesthetic dermatology, 8(12), 12–14.
2. Staples, M. P., Elwood, M., Burton, R. C., Williams, J. L., Marks, R., & Giles, G. G. (2006). Non-melanoma skin cancer in Australia: the 2002 national survey and trends since 1985. The Medical journal of Australia, 184(1), 6–10.
3. Sensus Healthcare. (2018). SRT-100+ TM Clinical Applications Handbook. Boca Raton, FL. CLN-SRT-051 R5.0, 82-88
4. David C. V. (2013). Electronic brachytherapy and superficial radiation therapy: will you be adding it to your practice?. Cutis, 92(5), E16–E18.
5. Roth, W. I., Shelling, M., & Fishman, K. (2019). Superficial Radiation Therapy: A Viable Nonsurgical Option for Treating Basal and Squamous Cell Carcinoma of the Lower Extremities. Journal of drugs in dermatology : JDD, 18(2), 130–134.
6. Nestor, M. S., Berman, B., Goldberg, D., Cognetta, A. B., Jr, Gold, M., Roth, W., Cockerell, C. J., & Glick, B. (2019). Consensus Guidelines on the Use of Superficial Radiation Therapy for Treating Nonmelanoma Skin Cancers and Keloids. The Journal of clinical and aesthetic dermatology, 12(2), 12–18.
7. Bhatnagar, A., & Loper, A. (2010). The initial experience of electronic brachytherapy for the treatment of non-melanoma skin cancer. Radiation oncology (London, England), 5, 87. https://doi.org/10.1186/1748-717X-5-87
8. Brewer, C. F., Deutsch, C. J., & Jemec, B. (2020). Is radiation therapy as a primary treatment modality for squamous cell carcinoma of the hand the best choice? Case series and review of the literature. Dermatology online journal, 26(6), 13030/qt0mx961gq.
9. Cognetta, A. B., Howard, B. M., Heaton, H. P., Stoddard, E. R., Hong, H. G., & Green, W. H. (2012). Superficial x-ray in the treatment of basal and squamous cell carcinomas: a viable option in select patients. Journal of the American Academy of Dermatology, 67(6), 1235–1241.
10. Petit, J. Y., Avril, M. F., Margulis, A., Chassagne, D., Gerbaulet, A., Duvillard, P., Auperin, A., & Rietjens, M. (2000). Evaluation of cosmetic results of a randomized trial comparing surgery and radiotherapy in the treatment of basal cell carcinoma of the face. Plastic and reconstructive surgery, 105(7), 2544–2551.
11. Wei, Q., Matanoski, G. M., Farmer, E. R., Hedayati, M. A., & Grossman, L. (1993). DNA repair and aging in basal cell carcinoma: a molecular epidemiology study. Proceedings of the National Academy of Sciences of the United States of America, 90(4), 1614–1618. https://doi.org/10.1073/pnas.90.4.1614











 Incredible Marketing
Incredible Marketing